By The TENS Magazine Editorial Staff
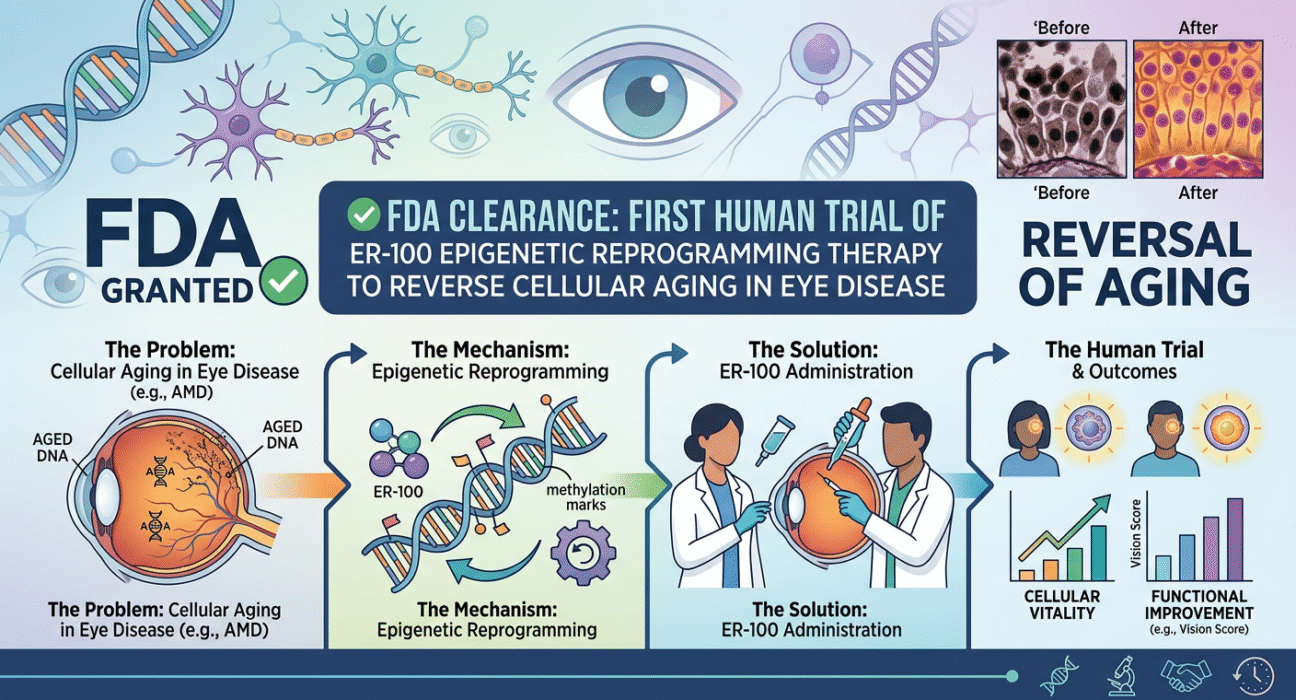
1. Regulatory milestone achieved The United States Food and Drug Administration has officially granted clearance for the first human clinical trial of ER-100, an investigational epigenetic reprogramming therapy. This regulatory decision allows researchers to transition from laboratory and animal testing into Phase 1 clinical trials involving human subjects. The clearance of an Investigational New Drug application indicates that the regulatory agency has reviewed the preclinical safety and efficacy data and determined that the proposed human trials meet the necessary ethical and scientific standards to proceed safely.
2. Mechanism of epigenetic reprogramming The ER-100 therapy utilizes the biological process of epigenetic reprogramming to alter how cells express their genetic instructions. Rather than changing the underlying DNA sequence, this approach modifies the chemical markers on the DNA that dictate cell behavior. By delivering specific transcription factors, the therapy aims to reset the epigenetic markers of older or damaged cells to a more youthful state. This process is designed to restore cellular function and promote tissue regeneration in areas where aging or disease has caused significant cellular degradation.
3. Strategic focus on ophthalmology The initial clinical application of ER-100 targets eye disease, a strategic choice in the field of gene and cellular therapy. The human eye is an ideal environment for testing novel genetic treatments because it is an immune-privileged site, meaning it has a reduced immune response compared to other parts of the body. Additionally, the eye is easily accessible for localized treatment administration, and clinicians can precisely measure structural and functional changes in the retina and optic nerve using non-invasive imaging technologies.
4. Reversing cellular aging in the retina The primary objective of the ER-100 therapy is to reverse cellular aging within the specific tissues of the eye, particularly targeting retinal ganglion cells. In many degenerative eye diseases, these cells lose their ability to function and communicate with the brain, leading to progressive vision loss. By resetting the epigenetic age of these cells, the therapy seeks to restore their youthful resilience and functional capacity, potentially allowing the cells to repair damage that is typically considered irreversible in adult human tissues.
5. Preclinical research foundation The FDA clearance for human trials is based on a comprehensive foundation of preclinical research. Prior to this approval, scientists conducted extensive in vitro studies and in vivo testing on animal models, including rodents and non-human primates. These preclinical studies were necessary to demonstrate that the epigenetic reprogramming factors could be safely delivered to the target cells, successfully initiate the reprogramming process without causing uncontrolled cell growth or tumor formation, and produce a measurable improvement in cellular function.
6. Phase one clinical trial objectives As a Phase 1 clinical trial, the initial human studies for ER-100 will prioritize the evaluation of safety and tolerability rather than therapeutic efficacy. Researchers will administer the therapy to a small, carefully selected cohort of patients suffering from specific degenerative eye conditions. The primary endpoints of this phase will involve monitoring patients for adverse immune reactions, systemic toxicity, and localized side effects within the eye, while also determining the optimal dosage required to achieve epigenetic changes safely.
7. Method of therapeutic administration The ER-100 therapy will likely be administered directly into the patient’s eye using an intravitreal injection, a standard delivery method for ophthalmic gene therapies. This localized approach ensures that the reprogramming factors reach the targeted retinal cells in high concentrations while minimizing systemic exposure to the rest of the body. The procedure is typically performed in a clinical setting under local anesthesia, allowing the therapeutic agents to penetrate the vitreous humor and access the damaged neural tissues at the back of the eye.
8. Implications for optic neuropathies The successful development of an epigenetic reprogramming therapy could fundamentally alter the treatment landscape for optic neuropathies, including conditions like glaucoma. Currently, treatments for these diseases focus primarily on slowing disease progression, such as lowering intraocular pressure, but they cannot restore lost vision. If ER-100 proves successful in reversing cellular aging and regenerating optic nerve function, it would represent a paradigm shift from disease management to restorative medicine for patients with degenerative vision loss.
9. Viral vector delivery systems To successfully deliver the epigenetic reprogramming instructions to the retinal cells, ER-100 relies on advanced delivery vehicle technology. Gene therapies typically utilize engineered viral vectors, such as adeno-associated viruses, which have been modified to remove their infectious properties. These vectors act as microscopic delivery trucks, carrying the therapeutic genetic material directly into the nucleus of the target cells. The FDA closely evaluates the safety profile of these vectors to ensure they do not trigger severe inflammatory responses in human subjects.
10. Timeline for clinical development The initiation of human trials marks the beginning of a multi-year clinical development process. Following the completion of the Phase 1 safety trial, researchers will need to conduct Phase 2 and Phase 3 trials to evaluate the therapy’s efficacy across larger and more diverse patient populations. Gathering conclusive data on visual acuity improvements and long-term safety will require extensive monitoring. Consequently, while the FDA clearance is a significant scientific milestone, widespread commercial availability of ER-100 remains several years away, pending successful outcomes in all clinical phases.