By The TENS Magazine Editorial Staff
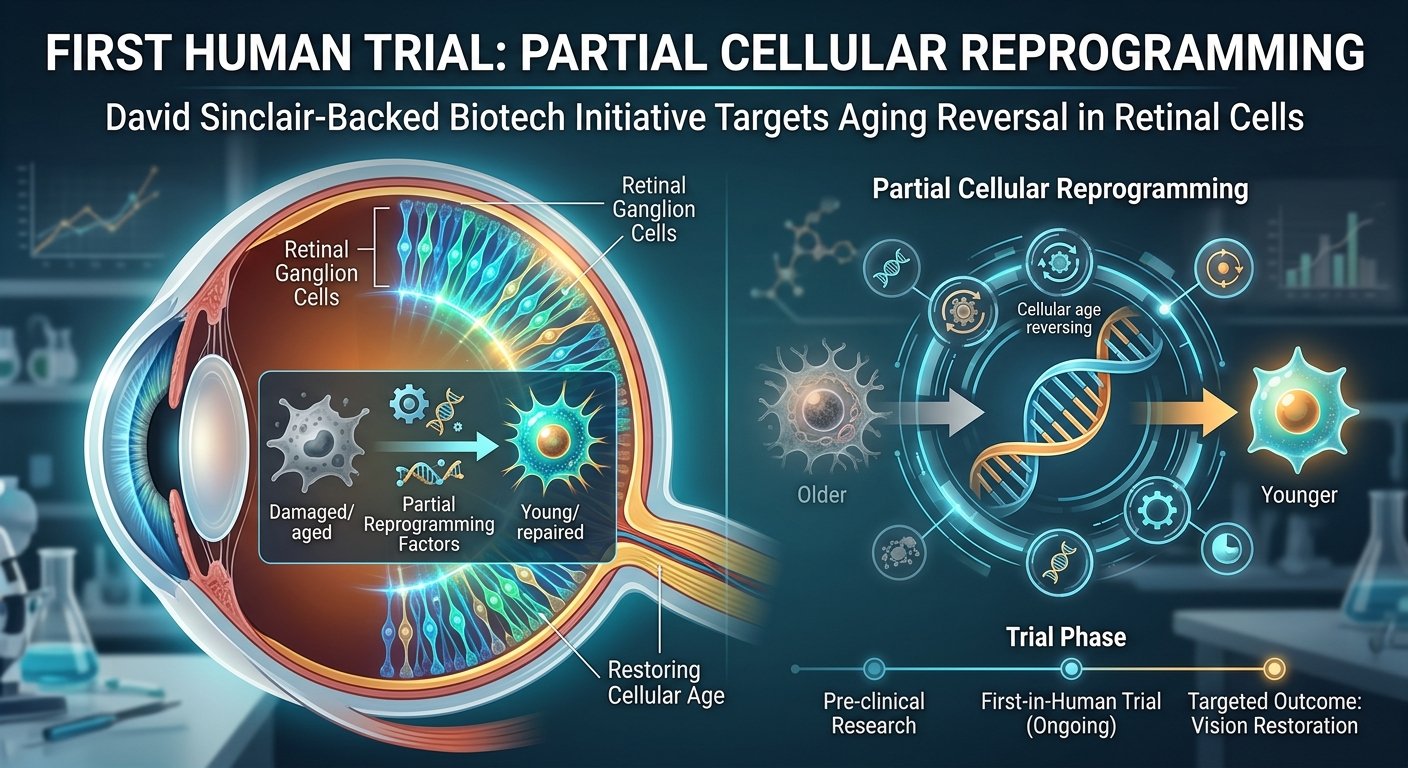
1. Core Technology The clinical trial utilizes partial cellular reprogramming, a biological process designed to reverse the aging of cells. This technique involves expressing specific genes known to induce pluripotency, but only for a limited duration. By doing so, scientists aim to restore cellular function without causing the cells to lose their original identity or form tumors.
2. Target Area The initial human trials focus specifically on retinal cells, which are critical for vision. The retina is an extension of the central nervous system, making it a practical candidate for testing neural regeneration. Researchers are targeting conditions associated with age-related vision loss, aiming to repair damaged optic nerves and restore visual acuity in patients.
3. Yamanaka Factors The reprogramming process relies on a subset of the Yamanaka factors, a group of proteins traditionally used to convert adult cells into stem cells. In this specific therapeutic application, researchers typically use three of the four original factors to safely alter the epigenetic markers of the retinal cells, avoiding the complete dedifferentiation that would erase the specialized function of the cell.
4. Role of Epigenetics The underlying science focuses on the epigenome, which controls how genes are turned on or off. As cells age, their epigenetic markers accumulate errors, leading to decreased function and cellular senescence. The partial reprogramming therapy seeks to remove these age-related epigenetic errors, effectively returning the retinal cells to a healthier biological state to improve overall tissue function.
5. Institutional Backing The biotechnology company conducting the trial is backed by David Sinclair, a researcher in the field of aging and genetics. Academic work previously conducted at Harvard Medical School laid the foundational research for this trial, demonstrating in animal models that epigenetic reprogramming could successfully restore vision in subjects with damaged optic nerves and age-related blindness.
6. Preclinical Testing Before advancing to human trials, the therapy underwent extensive preclinical testing in animal models, including mice and non-human primates. These studies demonstrated that the partial reprogramming technique could safely promote nerve regeneration and reverse vision loss caused by glaucoma and aging. The data from these preclinical models provided the necessary safety and efficacy metrics to secure regulatory approval for human testing.
7. Delivery Mechanism The genetic instructions required for partial cellular reprogramming are typically delivered to the retina using an adeno-associated virus vector. This viral vector is engineered to be harmless and serves solely as a delivery vehicle, transporting the therapeutic genes directly into the target cells of the eye. Once inside, the genes are activated to begin the epigenetic modification process.
8. Clinical Objectives The primary objective of this phase of the human clinical trial is to evaluate the safety and tolerability of the gene therapy in human subjects. While researchers will also monitor for signs of efficacy, such as improvements in visual function or optic nerve health, establishing a definitive safety profile is the critical first requirement in the regulatory approval process.
9. Broader Implications If the clinical trial proves successful, the implications extend beyond treating vision loss. Validating partial cellular reprogramming in humans would provide a proof of concept for treating a wide range of age-related diseases. The ability to safely reverse cellular aging in vivo could eventually lead to therapies targeting neurodegeneration, cardiovascular disease, and other conditions driven by cellular senescence.
10. Regulatory Oversight The initiation of this human trial requires rigorous regulatory and ethical oversight. Health authorities closely monitor the trial to ensure patient safety, particularly given the novel nature of gene therapy and cellular reprogramming. The data generated from this study will be subject to peer review and will help establish future regulatory frameworks for epigenetic medicines and related treatments.