By The TENS Magazine Editorial Staff
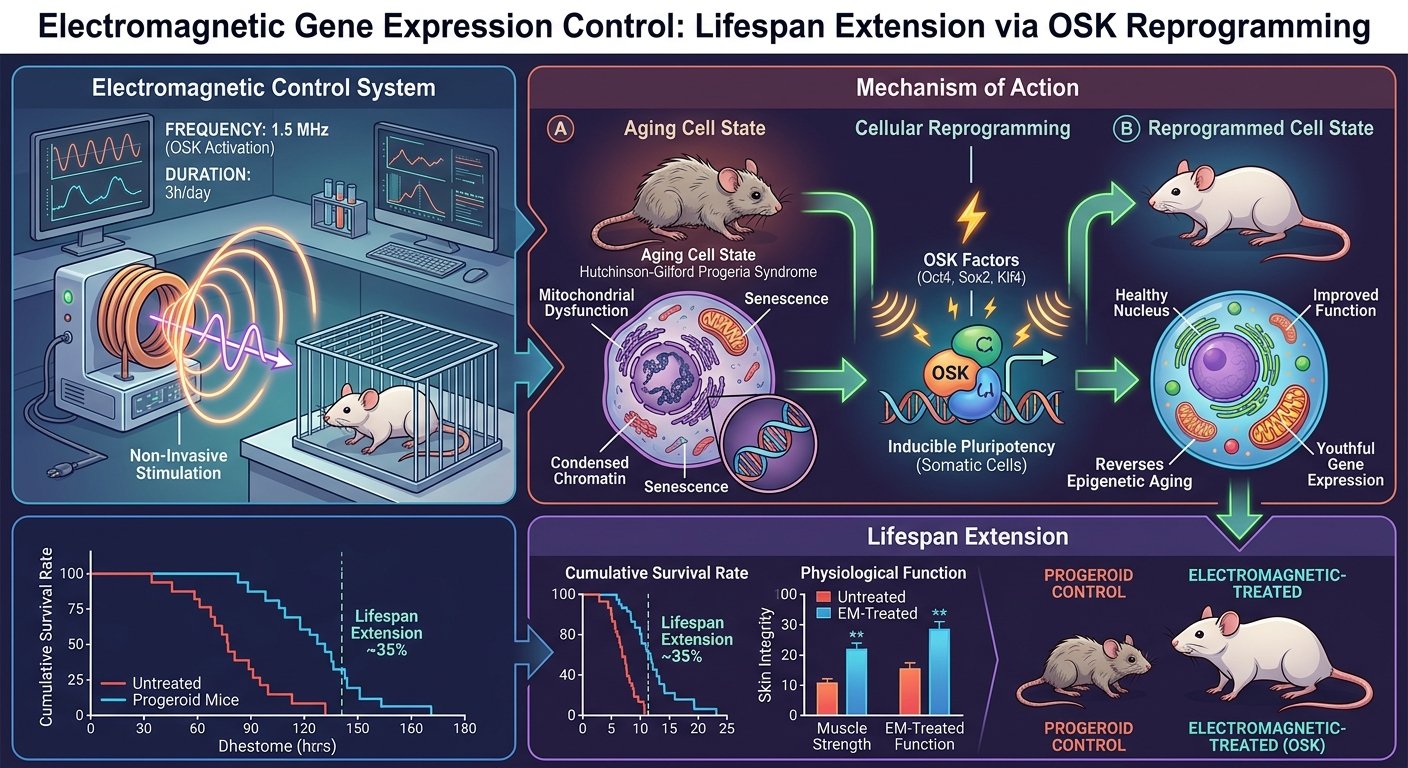
In a major advancement for genetic medicine and longevity research, scientists at Seoul National University have successfully demonstrated a non-invasive method to control gene expression using electromagnetic fields, resulting in significant lifespan extension in progeroid mice. Detailed in a recent publication in the journal Cell, the breakthrough utilizes a novel electromagnetic-responsive gene switch to remotely activate OSK (Oct4, Sox2, and Klf4) reprogramming factors. This innovative approach allows researchers to trigger partial epigenetic reprogramming—a process that reverses cellular aging markers—without relying on continuous drug administration, chemical inducers, or permanent viral implants. By applying specific 60 Hz electromagnetic pulses, the research team achieved temporal control over cellular rejuvenation, marking a critical step forward in the development of on-demand, externally controlled gene therapies.
At the core of this technological leap is an engineered electromagnetic-field-inducible promoter element, designated by the researchers as Ei. To understand how magnetic fields can manipulate cellular functions, the team conducted extensive CRISPR screening to identify the molecular pathways responsive to these specific frequencies. They discovered that a protein known as CYB5B acts as a biological sensor for rhythmic calcium oscillations generated by the external electromagnetic pulses. When exposed to a precise condition of 2.0 mT (millitesla) at 60 Hz, CYB5B detects the temporal rhythm of the calcium shifts and subsequently triggers the transcription factor SP7 to activate the target genes.
Crucially, the system is highly specific; it responds exclusively to the rhythmic oscillatory pattern rather than random or non-specific increases in cellular calcium intensity. This mechanism functions as a biological lock that only opens when presented with the correct electromagnetic input. The selection of the 60 Hz frequency is particularly notable, as it aligns with standard alternating current (AC) power frequencies utilized in many parts of the world, suggesting that future therapeutic hardware could potentially be adapted from existing electrical infrastructure principles.
The therapeutic payload delivered by this electromagnetic switch relies on the well-documented Yamanaka factors, specifically the OSK subset, which excludes the oncogenic c-Myc gene to minimize cancer risks. Partial cellular reprogramming via OSK expression has previously been shown to restore youthful epigenetic markers and improve tissue function. However, a persistent challenge in the field of longevity research has been controlling the dosage and duration of this reprogramming. Continuous or excessive expression of these factors can lead to cellular dedifferentiation, tissue dysplasia, and increased mortality.
The Seoul National University team utilized the Ei switch to establish a highly controlled, cyclic regimen. Through rigorous optimization, they determined that a schedule of three days of electromagnetic activation followed by four days of rest (3 days ON / 4 days OFF) provided the optimal balance. This cyclic Ei-OSK induction allowed the cells to undergo rejuvenation without losing their original cellular identity, reinforcing the scientific consensus that timing and dosage are the most critical variables in epigenetic reprogramming.
To test the efficacy of the Ei-OSK system, the researchers utilized progeroid mice—a genetic model engineered to exhibit accelerated aging, mimicking conditions like Hutchinson-Gilford progeria syndrome. The treatment protocol for the progeroid cohort commenced at three months of age and continued for 90 days. The results were highly pronounced. Mice subjected to the cyclic electromagnetic treatment demonstrated a visibly improved physical appearance, a significant reduction in age-related spinal curvature (kyphosis), and a mitigation of the rapid body-weight decline typically associated with the progeroid condition.
Most importantly, the cyclic activation of the OSK genes resulted in a substantial extension of both the median and maximal lifespan of the treated mice compared to the control groups. Molecular and histological analyses confirmed these macroscopic observations. Tissues from the treated mice exhibited restored vascular structures, a reversal of age-linked histone methylation marks, and a marked decrease in the expression of p16INK4a, a widely recognized biomarker for cellular senescence. The researchers also replicated these regenerative effects in naturally aged wild-type mice, initiating treatment at 20 months of age for a duration of 120 days, further validating the broad applicability of the electromagnetic gene switch.
The implications of this study extend far beyond the realm of murine models. Currently, the majority of gene therapies require either permanent genetic modifications, which carry long-term risks, or the continuous administration of chemical inducers, which can have systemic side effects. The Ei system introduces the paradigm of remote, reversible, and temporal control over genetic expression. If this pathway is conserved in human cells, clinicians could theoretically activate regenerative or therapeutic genes on demand, and immediately halt their expression once the clinical objective is achieved.
Beyond anti-aging and longevity, the researchers demonstrated the versatility of the platform by engineering an inducible model of Alzheimer’s disease. By placing mutant humanized APP (amyloid precursor protein) variants under the control of the Ei promoter, they were able to trigger Alzheimer’s pathology in already-aged brains using localized electromagnetic fields. This isolates the effects of the disease from early developmental factors, providing a more accurate model for age-onset neurodegeneration.
Despite the groundbreaking nature of the findings, several hurdles remain before electromagnetic gene therapy can transition to human clinical trials. A primary concern is the penetration depth of electromagnetic fields in larger organisms. While the coils effectively stimulated the target tissues in mice, human organs are situated much deeper, and surrounding tissues may attenuate or distort the magnetic fields. Furthermore, the long-term safety of chronically activating cellular calcium signaling pathways via CYB5B requires comprehensive investigation in larger animal models. While 60 Hz fields are generally considered safe for brief exposures, the physiological impact of repeated, long-term stimulation remains an open question.
Nevertheless, the publication in Cell establishes a robust proof of concept. The ability to engineer a biological system that responds to external, non-invasive electromagnetic signals with high genetic specificity represents a transformative milestone. As researchers continue to refine the technology and explore its applications, electromagnetic gene expression control could eventually become a cornerstone of next-generation regenerative medicine, offering unprecedented precision in the fight against age-related diseases.